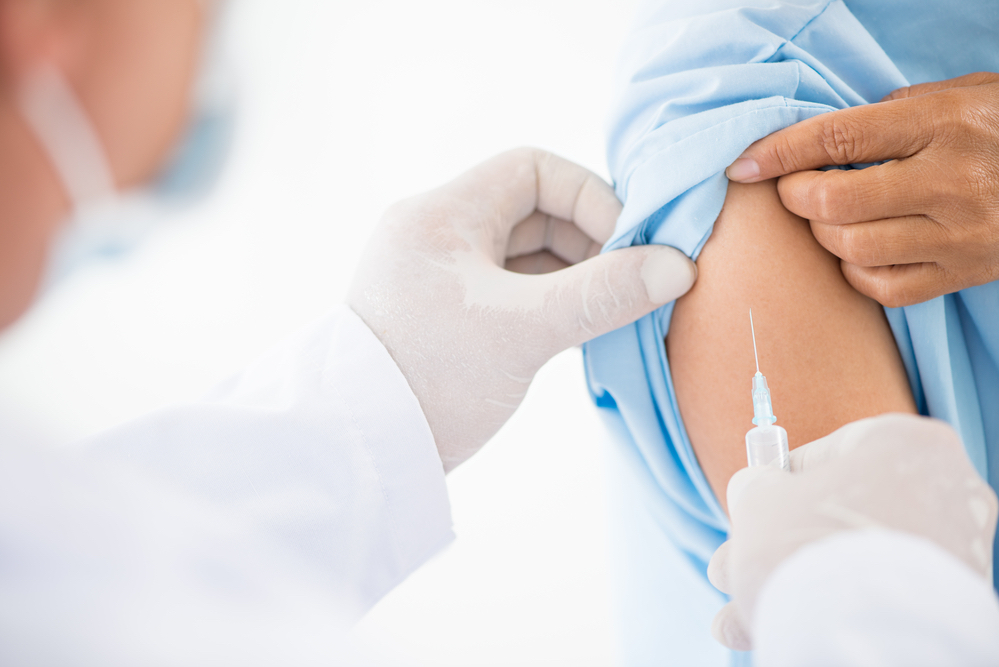
Now, as the Food and Drug Administration (FDA) has issued emergency authorization for two COVID-19 vaccines in the U.S. and rollouts have begun, you may have a lot of questions about what this means for you and the people you love.
It’s important to gain information from credible sources and talk with your healthcare provider to make a plan that’s right for you.
Over the coming weeks I will be answering more questions but for starters, here’s some key points:
- Yes, COVID-19 vaccine development has been fast — but researchers haven’t cut corners when it comes to safety. Through projects like Operation Warp Speed in the U.S., and global commitment, the COVID-19 vaccine development process has been “supercharged” for developing effective vaccines. Thousands of researchers have been focused on vaccine development. Clinical trials obtained eager participants, allowing it to advance quickly, but carefully.
- The FDA — the gold-standard drug and vaccine regulatory authority in the world —is staffed by scientists and non-political appointees, whose chief concern is the health and safety of the American people. They have intensively studied data from the companies before providing Emergency Use Authorization (EUA).
- Pfizer and Moderna have both shown the vaccines to be highly effective, with a strong safety profile – and they cannot give a person COVID-19.
- With wide distribution and vaccinations, we could crush this outbreak similar to what we did with smallpox, polio and measles.
Who should get this vaccine?
- I join with millions of physicians and health experts across our country in urging everyone to make a vaccination plan — consult with your healthcare provider, learn when and where you may be able to access a vaccine, and how many doses you will take based on the vaccine formula as well as any related side effects that may affect you.
- Seek information from reliable sources
- Beware of misinformation that thrives on social media
- Consider too that when you make the decision, you aren’t just helping yourself — but also protecting the most vulnerable people in your circle.
How long does the COVID vaccine last, once administered?
- Natural immunity to COVID-19, meaning the protection an individual gains from already having been infected, varies from person-to-person. The CDC warns that cases of reinfection have been reported. Experts advise that people who have gotten sick with COVID-19 in the past should still get the vaccine due to risk of reinfection.
- Both Moderna and Pfizer announced that their COVID-19 vaccines have around 95% efficacy. The vaccines could provide long-lasting protection or fade away in under a year and require a booster. Scientists/researchers will be evaluating the vaccine’s duration.
Can employers force employees to get the vaccine?
- Generally, employers can require their employers to get vaccines as a condition of employment if it is job-related and considered a business necessity, legal experts say. The idea is to keep the workplace safe. However, few are expected to, as there is understanding that many nuances exist and it’s important to manage with caution.
- That being said, America’s best-line of defense against COVID-19 depends on folks rolling up their sleeves, in a new way. Vaccines have been proven safe with a long, scientifically-proven body of work in protecting people.
- Employers should be planning how they will handle vaccine information, communications and any related services or processes.
According to surveys, some people are leery of the new vaccines and their safety. . .
- Vaccines don’t save lives, vaccinations do and there is a lot of misinformation and disinformation out there. Our physicians and pharmacists leading this rollout have full confidence in the vaccine’s safety and the science behind them.
- Vaccine misinformation has grown exponentially with the internet and social media, fueling the anti-vaxxer movement to the point of creating hesitancy across this country.
- Diminishing the reluctance won’t be easy, experts say. It’s entangled in decades-old hesitance and fears of unknown side effects, combined with the speed the COVID-19 vaccine was developed.
- We must responsibly address concerns by educating the facts.
How about children?
- Infectious disease experts recently announced that once vaccines hit the market, they will not initially be available to children because few children have participated in clinical trials for the vaccine. Children’s immune systems are different from adults. As such, there might be discrepancies in vaccine effectiveness between the age groups.
- Clinical trials with children must be implemented and the child vaccine protocol must be approved by the FDA.
We must not let up on practicing basic public health measures like mask-wearing, physical distancing, and handwashing. But now is the time for everyone to prepare for the arrival of vaccine options — by getting educated, making plans to be vaccinated, and understanding the importance of widespread vaccine uptake if we are to ever to crush COVID-19 and return to pre-pandemic life.
Dr. Nina Radcliff is dedicated to her profession, her patients and her community, at large. She is passionate about sharing truths for healthy, balanced living as well as wise preventive health measures.
She completed medical school and residency training at UCLA and has served on the medical faculty at The University of Pennsylvania. She is a Board Certified Anesthesiologist. Author of more than 200 textbook chapters, research articles, medical opinions and reviews; she is often called upon by media to speak on medical, fitness, nutrition, and healthy lifestyle topics impacting our lives, today.
Dr. Nina Radcliff is dedicated to her profession, her patients and her community, at large. She is passionate about sharing truths for healthy, balanced living as well as wise preventive health measures.
She completed medical school and residency training at UCLA and has served on the medical faculty at The University of Pennsylvania. She is a Board Certified Anesthesiologist. Author of more than 200 textbook chapters, research articles, medical opinions and reviews; she is often called upon by media to speak on medical, fitness, nutrition, and healthy lifestyle topics impacting our lives, today.